The use of carbon in the form of charcoal has been used for many applications. Even in Hindu documents dating from 450 BC charcoal filters are mentioned for the treatment of water. Charred wood, bones and coconut charcoals were used during the 18th and 19th century by the sugar industry for decolourising solutions. Activated carbon is a material prepared in such a way that it exhibits a high degree of porosity and an extended surface area.
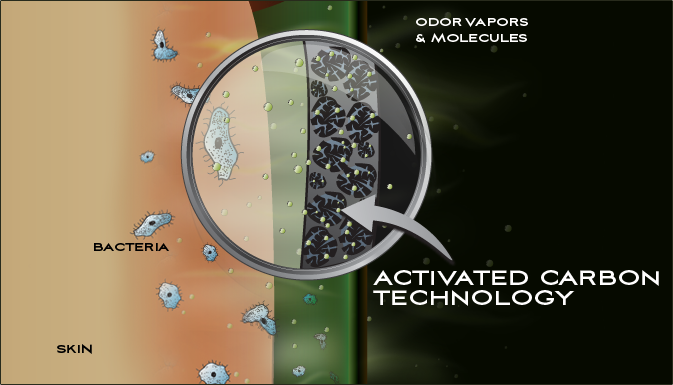
During water filtration through activated carbon, contaminants become trapped in the small pores of the activated carbon. This process is called adsorption. Activated carbon filters are efficient to remove certain organics (such as unwanted taste and odours, micropollutants), chlorine, fluorine or radon, from drinking water or wastewater.
Treatment Principles
There are two basic types of water filters: particulate filters and adsorptive/reactive filters. Particulate filters exclude particles by size, and adsorptive/reactive filters contain a material (medium) that either adsorbs or reacts with a contaminant in water. The principles of adsorptive activated carbon filtration are the same as those of any other adsorption material. The contaminant is attracted to and held (adsorbed) on the surface of the carbon particles. The characteristics of the carbon material (particle and pore size, surface area, surface chemistry, etc.) influence the efficiency of adsorption.
The characteristics of the chemical contaminant are also important. Compounds that are less water-soluble are more likely to be adsorbed to a solid. A second characteristic is the affinity that a given contaminant has with the carbon surface. This affinity depends on the charge and is higher for molecules possessing less charge. If several compounds are present in the water, strong adsorbents will attach to the carbon in greater quantity than those with weak adsorbing ability.

Raucherkabine Outdoor
Eine Raucherkabinen für den Außenbereich bietet mehr Vorteile als die Reinigung der Luft. Darüber hinaus garantieren unsere Lösungen eine effektive Beseitigung von Gasen, Gerüchen und anderen gesundheitsschädlichen Partikeln.
Die Gründe sind zahlreich und um mehr zu erfahren, kontaktieren Sie uns bitte noch heute. Unsere Outdoor Raucherkabinen bieten sichere, komfortable Räume für mehrere Raucher gleichzeitig, ohne dass die Umgebung durch Passivrauchen beeinträchtigt wird.